What Are Gadolinium-Based Contrast Agents (GBCAs)?
Gadolinium-based contrast agents (GBCAs) are intravenous substances used during magnetic resonance imaging (MRI) scans to improve the visibility of internal structures. Approximately 30 million contrast-enhanced MRI scans are performed worldwide each year, and GBCAs are the standard contrast medium used in the vast majority of these procedures. They are routinely administered to visualize blood vessels, detect tumors, identify areas of inflammation, assess organ function, and evaluate the integrity of the blood-brain barrier.
The active element in every GBCA is gadolinium, a rare earth metal with the atomic number 64. Gadolinium possesses unique paramagnetic properties — it has seven unpaired electrons, more than any other element — which allow it to dramatically alter the magnetic relaxation times of nearby water molecules. When a GBCA enters the bloodstream and reaches the tissue being imaged, the gadolinium ions shorten the T1 relaxation time of surrounding water protons, causing those areas to appear brighter on T1-weighted MRI images. This enhanced contrast helps radiologists distinguish between normal and abnormal tissue, detect small lesions that would otherwise be invisible, and assess the vascularity of tumors and other structures.
In its elemental or free ionic form, gadolinium is highly toxic to biological systems. To make it safe for injection into patients, gadolinium is bound to a chelating molecule — a specially designed organic ligand that wraps around the gadolinium ion and prevents it from interacting with body tissues. The chelate is intended to keep the gadolinium stable during imaging and facilitate its excretion through the kidneys, ideally within 24 to 48 hours in patients with normal renal function. However, as research since 2014 has demonstrated, this excretion is not always complete, and gadolinium can be retained in the body for months, years, or even decades after exposure.
The safety profile of a GBCA depends heavily on the design of its chelating molecule. Not all chelates hold gadolinium with equal strength, and this difference is the foundation of the most important distinction in GBCA classification: the difference between linear and macrocyclic agents. Understanding this distinction is essential for patients, referring physicians, and radiologists when evaluating the risks and benefits of contrast-enhanced MRI.
Linear vs. Macrocyclic Gadolinium Agents
The most clinically significant difference between GBCAs is the structure of the chelating molecule that holds the gadolinium ion. This structural difference directly impacts the agent's thermodynamic stability, kinetic inertness, and the likelihood that gadolinium will dissociate from the chelate and deposit in tissues. All approved GBCAs fall into one of two structural categories: linear or macrocyclic.
Linear Agents (Open-Chain)
Linear GBCAs use an open-chain chelating molecule that wraps around the gadolinium ion but does not fully enclose it. This structure provides less thermodynamic stability and kinetic protection, meaning the gadolinium ion can more easily dissociate from the chelate through a process called transmetallation.
- •Open-chain molecular structure
- •Lower thermodynamic stability
- •Higher rates of gadolinium dissociation
- •Greater susceptibility to transmetallation
- •Higher levels of gadolinium retention in tissues
- •Several agents suspended or restricted in the EU
- •Associated with more cases of brain signal changes on MRI
Macrocyclic Agents (Cage-Like)
Macrocyclic GBCAs use a cage-like or ring-shaped chelating molecule that fully encloses the gadolinium ion in a pre-organized cavity. This structure provides significantly greater thermodynamic stability and kinetic inertness, meaning the gadolinium is much less likely to be released into the body.
- •Closed cage-like molecular structure
- •Higher thermodynamic stability
- •Lower rates of gadolinium dissociation
- •Greater resistance to transmetallation
- •Lower levels of gadolinium retention in tissues
- •All macrocyclic agents remain approved worldwide
- •Preferred by regulatory agencies when contrast is necessary
It is important to note that while macrocyclic agents are considered safer than linear agents, they are not risk-free. Research has documented gadolinium retention even from macrocyclic GBCAs, and published patient survey data shows that the same 14 nervous system symptoms ranked in the top 25 for patients who received only linear GBCAs and those who received only macrocyclic GBCAs. This suggests that the underlying mechanism of gadolinium toxicity can occur with any type of GBCA, though the risk appears to be lower with macrocyclic formulations.
| Feature | Linear Agents | Macrocyclic Agents |
|---|---|---|
| Molecular Structure | Open chain | Closed cage / ring |
| Thermodynamic Stability | Lower | Higher |
| Kinetic Inertness | Lower | Higher |
| Gd Release Risk | Higher (more transmetallation) | Lower (more stable) |
| Brain Retention | Higher (demonstrated on T1 MRI) | Lower (but still documented) |
| Regulatory Status (EU) | Several suspended (2017) | All remain approved |
| Examples | Magnevist, Omniscan, OptiMARK | Dotarem, ProHance, Gadavist |
All Approved Gadolinium-Based Contrast Agents
The following is a comprehensive list of the gadolinium-based contrast agents that have been approved for clinical use. Each agent uses a different chelating molecule with distinct stability characteristics. Understanding which agent you received — or which agent is being proposed for an upcoming MRI — is an important part of evaluating your personal risk profile. The specific GBCA used should be documented in your radiology report and medical records.
Linear Agents (Higher Risk of Gadolinium Release)
These agents use an open-chain chelate structure and are associated with higher rates of gadolinium dissociation, tissue retention, and brain signal changes on MRI. Several have been suspended or restricted in the European Union.
| Brand Name | Generic Name | Manufacturer | Type | Charge |
|---|---|---|---|---|
| Magnevist | Gadopentetate dimeglumine | Bayer | Linear | Ionic |
| Omniscan | Gadodiamide | GE Healthcare | Linear | Nonionic |
| OptiMARK | Gadoversetamide | Guerbet | Linear | Nonionic |
| MultiHance | Gadobenate dimeglumine | Bracco | Linear | Ionic |
Magnevist was one of the first GBCAs approved for clinical use (1988) and was widely used for decades. Omniscan is a nonionic linear agent that has been particularly associated with Nephrogenic Systemic Fibrosis (NSF) in patients with renal impairment. Both Omniscan and Magnevist were among the agents whose intravenous formulations were suspended by the EMA in 2017 for standard body imaging. MultiHance has a partial hepatobiliary excretion pathway, which is why it retains a limited indication for liver imaging in the EU.
Macrocyclic Agents (Lower Risk of Gadolinium Release)
These agents use a cage-like chelate structure that more securely encloses the gadolinium ion. They are associated with lower rates of gadolinium release, lower tissue retention, and remain fully approved in all major regulatory jurisdictions.
| Brand Name | Generic Name | Manufacturer | Type | Charge |
|---|---|---|---|---|
| Dotarem / Clariscan | Gadoterate meglumine | Guerbet | Macrocyclic | Ionic |
| ProHance | Gadoteridol | Bracco | Macrocyclic | Nonionic |
| Gadavist / Gadovist | Gadobutrol | Bayer | Macrocyclic | Nonionic |
Dotarem (also marketed as Clariscan) is an ionic macrocyclic agent with one of the highest thermodynamic stability constants among all GBCAs. ProHance was one of the first macrocyclic agents approved and has a long safety record. Gadavist (known as Gadovist outside the US) is a high-concentration macrocyclic agent that allows for smaller injection volumes. All three macrocyclic agents remain fully approved for use in the United States, European Union, and other major markets.
Transmetallation: How Gadolinium Escapes the Chelate
Transmetallation is the central chemical process underlying gadolinium toxicity concerns. It refers to the exchange reaction in which endogenous metal ions present in the body — particularly zinc (Zn2+), copper (Cu2+), calcium (Ca2+), and iron (Fe3+) — compete with gadolinium for binding sites on the chelating molecule. When an endogenous metal successfully displaces gadolinium from the chelate, the gadolinium ion is released in its free, unchelated form (Gd3+), and the endogenous metal takes its place within the chelate structure.
This process is governed by thermodynamic stability constants and kinetic inertness. A chelate with high thermodynamic stability has a strong binding affinity for gadolinium, making it energetically unfavorable for other metals to displace it. A chelate with high kinetic inertness releases gadolinium slowly, even under conditions that thermodynamically favor release. Macrocyclic agents excel on both measures: their pre-organized cage structure both binds gadolinium tightly and resists conformational changes that would allow the ion to escape. Linear agents, with their more flexible open-chain structure, are inherently more vulnerable to both thermodynamic displacement and kinetic release.
The biological consequences of transmetallation are significant. When zinc is used to displace gadolinium from the chelate, the body's zinc stores can be depleted. Zinc is an essential trace mineral involved in immune function, wound healing, protein synthesis, and hundreds of enzymatic reactions. Some researchers have proposed that zinc depletion from transmetallation may contribute to the immune dysregulation and other symptoms observed in patients with gadolinium retention. Meanwhile, the free Gd3+ ions released through transmetallation can deposit in various tissues, triggering the cascade of toxic effects described in the following section.
In vitro studies have demonstrated that transmetallation rates vary substantially among GBCAs. Omniscan (gadodiamide) and Magnevist (gadopentetate dimeglumine) consistently show the highest rates of gadolinium release in competitive binding assays with zinc and other metals. Macrocyclic agents like Dotarem (gadoterate meglumine) show minimal gadolinium release under the same conditions. These laboratory findings are consistent with the clinical observations that linear agents are associated with higher levels of gadolinium retention in patient tissues, including the brain.
Free Gadolinium: Why Unchelated Gd3+ Is Toxic
When gadolinium dissociates from its chelating molecule, the resulting free gadolinium ion (Gd3+) is a potent biological toxin. The FDA noted in 2007that "free gadolinium is toxic" and specifically cited its effects on the liver and its interference with calcium channels. Understanding why free gadolinium is harmful requires examining its chemical properties and how they interact with normal biological processes.
The toxicity of free gadolinium stems primarily from its similarity to calcium. The Gd3+ ion has an ionic radius of approximately 1.05 angstroms, which is remarkably close to the calcium ion (Ca2+) radius of approximately 1.00 angstroms. Because of this size similarity, gadolinium can substitute for calcium in many biological processes — but with dramatically different effects. Gadolinium is a trivalent ion (3+ charge) compared to calcium's divalent charge (2+), meaning it binds more tightly to calcium-binding sites and can effectively block the normal function of voltage-gated calcium channels. These channels are critical for nerve signal transmission, muscle contraction, neurotransmitter release, and hormonal secretion.
The consequences of calcium channel disruption by gadolinium are wide-ranging. In the nervous system, it can impair signal conduction, potentially explaining the burning pain, tingling, numbness, and cognitive difficulties reported by patients with gadolinium retention. In muscle tissue, calcium channel interference can manifest as muscle twitching, spasms, and weakness. In the endocrine system, disrupted calcium signaling may affect thyroid function, hormonal balance, and metabolic regulation.
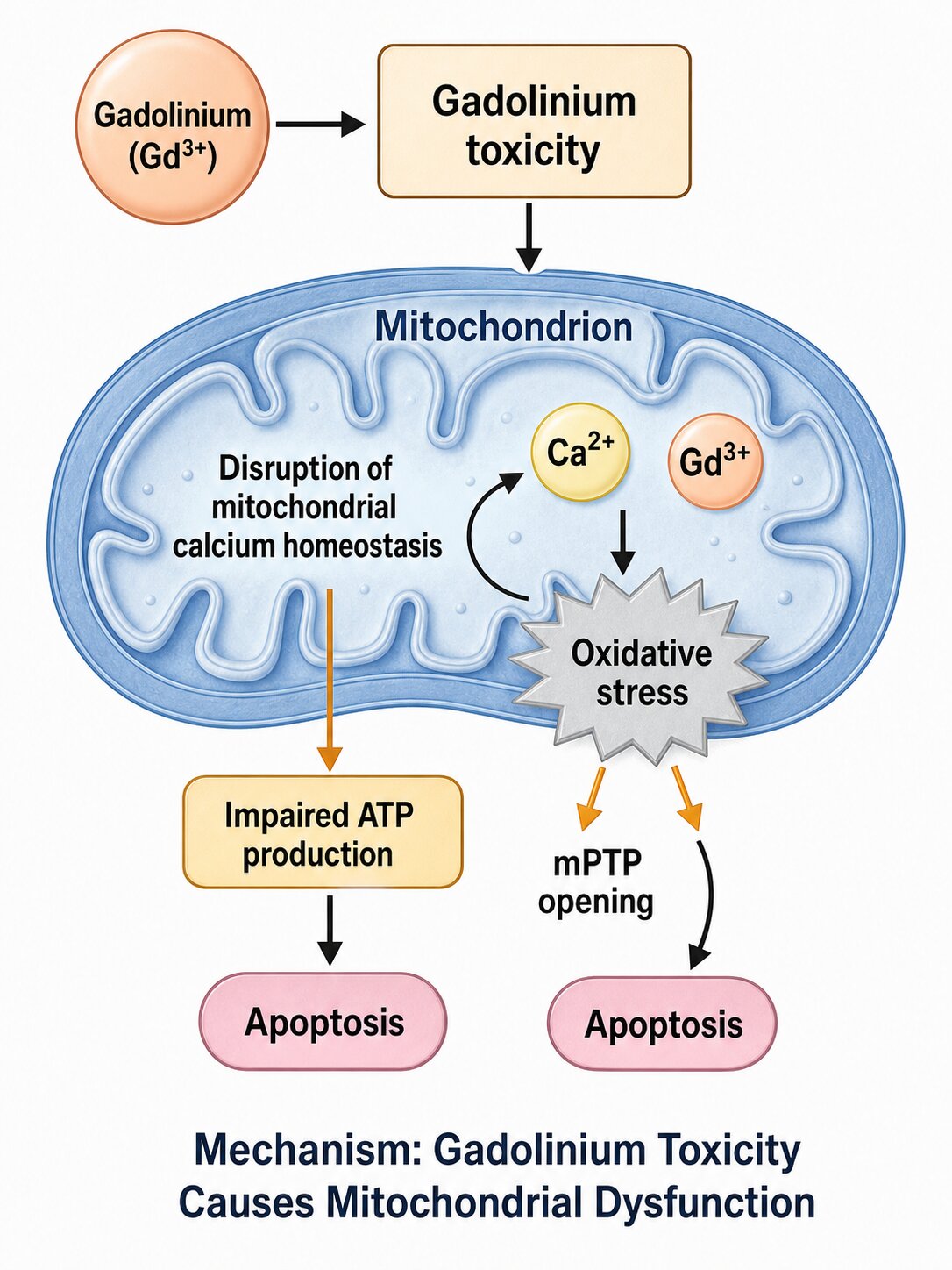
Research has demonstrated that free gadolinium deposits preferentially in certain tissues. In the brain, gadolinium accumulates most prominently in the dentate nucleus and globus pallidus, which appear as areas of increased signal intensity on unenhanced T1-weighted MRI scans. This finding, first published by Kanda et al. in 2014, was a landmark discovery because it proved that gadolinium crosses the blood-brain barrier and persists in brain tissue even in patients with normal kidney function. Beyond the brain, gadolinium has been detected in bone (where it may incorporate into the hydroxyapatite crystal structure), skin (contributing to the thickening and fibrosis seen in NSF), kidneys, liver, and various other organs.
Once deposited in tissues, free gadolinium can also trigger inflammatory and immune responses. The body recognizes the foreign metal ion as a threat and mounts an inflammatory cascade that can include activation of macrophages, release of pro-inflammatory cytokines, and stimulation of fibrotic pathways. This chronic inflammatory response is believed to be a key mechanism underlying the skin and connective tissue changes seen in NSF and may contribute to the systemic symptoms of Gadolinium Deposition Disease. Some researchers have also noted that gadolinium can form insoluble precipitates with phosphate ions in the body, creating gadolinium-phosphate deposits that act as long-term reservoirs of the toxic metal.
Regulatory Actions on Gadolinium Contrast Agents
Regulatory agencies worldwide have taken a series of increasingly significant actions regarding gadolinium contrast agents as evidence of retention and toxicity has accumulated. These actions reflect a growing recognition that the risks of certain GBCAs — particularly linear agents — extend beyond the previously known concern of Nephrogenic Systemic Fibrosis in patients with renal impairment.
European Medicines Agency (EMA) — 2017
In November 2017, the EMA's Pharmacovigilance Risk Assessment Committee (PRAC) recommended suspending the marketing authorizations for several intravenous linear GBCAs used in body scans. The suspended agents included Omniscan (gadodiamide), Magnevist (gadopentetate dimeglumine), and OptiMARK (gadoversetamide). The committee concluded that these linear agents lead to greater accumulation of gadolinium in brain tissue compared to macrocyclic agents, and that the long-term risks of brain deposition were not yet fully understood.
MultiHance (gadobenate dimeglumine) retained a restricted indication for liver imaging due to its hepatobiliary uptake properties, which provide diagnostic value not available from macrocyclic agents. The EMA's decision was the most decisive regulatory action taken globally and led to a significant shift toward macrocyclic agents in European clinical practice.
US Food and Drug Administration (FDA)
The FDA has taken a more cautious regulatory approach than the EMA. In 2007, the FDA added a boxed warning to all GBCA labels regarding the risk of NSF in patients with renal impairment. In 2017, the FDA required a new class warning for all GBCAs stating that gadolinium is retained in the body, including the brain, and issued a safety communication requiring that patients receive a Medication Guide before receiving a GBCA.
Unlike the EMA, the FDA did not suspend any linear agents, stating that the clinical consequences of brain gadolinium retention were not yet clear. However, the FDA's safety communication recommended that healthcare providers "consider the retention characteristics of each agent" when choosing a GBCA and noted that linear agents are associated with higher gadolinium retention. The FDA continues to monitor the issue and has stated it will update its guidance as new evidence becomes available.
Other Regulatory Bodies
Japan's Pharmaceuticals and Medical Devices Agency (PMDA) and Health Canada have both issued advisories about gadolinium retention, though neither has taken action as decisive as the EMA's suspension. Australia's Therapeutic Goods Administration (TGA) has also issued safety communications. The general trend across regulatory agencies has been to acknowledge the evidence of gadolinium retention, recommend preference for macrocyclic agents when clinically appropriate, and call for continued monitoring and research into the long-term consequences of tissue gadolinium deposition.
What This Means for Patients
If you have had one or more contrast-enhanced MRI scans, understanding which GBCA was used is an important step in evaluating your personal exposure history. Many patients who later develop symptoms of gadolinium retention do not initially know which contrast agent they received, because the choice of GBCA is typically made by the radiology department rather than the referring physician, and patients are rarely given this information proactively.
How to Find Out Which GBCA You Received
- Request your full radiology report from the imaging facility. The GBCA brand name, generic name, dose, and sometimes lot number should be documented in the contrast injection records or nursing notes.
- Contact the imaging center directly if the contrast agent is not listed in your radiology report. Facilities are required to maintain records of which contrast agents are administered to each patient.
- Check your hospital portal or patient records. Many healthcare systems now provide online access to imaging reports and procedure details.
- Ask before your next MRI. If a contrast-enhanced MRI is recommended, ask your physician and the radiology department which specific GBCA will be used and whether a macrocyclic agent is available.
Questions to Ask Your Radiologist
- Which specific GBCA will be used for my scan?
- Is a macrocyclic agent available at this facility?
- Is contrast truly necessary for this particular imaging study, or can adequate diagnostic information be obtained without it?
- What is the minimum effective dose of contrast for my scan?
- Will the brand name, dose, and lot number be documented in my records?
Keeping a personal record of every contrast-enhanced MRI you receive — including the date, imaging facility, specific GBCA used, and dose administered — is strongly recommended. This information is invaluable if you later need to assess your gadolinium exposure history, undergo testing for gadolinium retention, or consult with a physician experienced in gadolinium toxicity. For patients who have already received multiple contrast MRIs and are experiencing symptoms, obtaining this historical information is an important first step in building a complete clinical picture.
Frequently Asked Questions About Gadolinium Contrast Agents
What is the difference between macrocyclic and linear gadolinium contrast agents?▼
Which gadolinium contrast agent is the safest for MRI?▼
What is transmetallation and why does it matter for MRI contrast safety?▼
Why is free gadolinium toxic to the body?▼
How can I find out which gadolinium contrast agent was used during my MRI?▼
Where to Go Next
MRI Side Effects
Complete guide to contrast dye dangers
All Symptoms
Full symptom list from 316 patients
Deposition Disease
GDD symptoms, diagnosis, and treatment
Blood Test Guide
How to test for gadolinium levels
FAQ
Common questions about gadolinium
Find a Doctor
Physicians experienced with gadolinium
Sources and Review
Author: Gadolinium.org Editorial Team (Patient-Led Education)
Last reviewed: April 5, 2026
Medical review context: Content based on published peer-reviewed research, FDA safety communications, EMA regulatory decisions, and the 316-patient gadolinium retention survey.
This page is for education only and is not a diagnosis or treatment plan.
References
- FDA Drug Safety Communication: FDA warns that gadolinium-based contrast agents (GBCAs) are retained in the body
- EMA - PRAC concludes assessment of gadolinium agents used in MRI scans (2017)
- Kanda T, et al. High Signal Intensity in the Dentate Nucleus and Globus Pallidus on Unenhanced T1-weighted MR Images. Radiology. 2014.
- Survey of the Chronic Effects of Retained Gadolinium from Contrast MRIs (Patient Survey)